Research UPDATE: Using the Newest Technology to Understand Hydrocephalus
 By Dr. Ramin Eskandari, former Mentored Young Investigator for the Hydrocephalus Association
By Dr. Ramin Eskandari, former Mentored Young Investigator for the Hydrocephalus Association
Our understanding of infantile hydrocephalus pathophysiology (the study of abnormal biological processes which result in the signs and symptoms of a disease) contains many knowledge gaps, including insight into when and what regions deep within the brain are injured and how treatments affect this process. My recent studies, supported by the Hydrocephalus Association through the Mentored Young Investigators Award, have utilized some of the newest technologies in the world of brain imaging – Diffusion Tensor Imaging (DTI) on MRI – to answer these complex questions. In an early study, we were able to relate the timing of ventricular reservoir placement and ventricular size with neurological function in a hydrocephalus animal model. In our current study, we examined white matter tracts deep within the developing brain to see how susceptible each was to the effects of hydrocephalus.
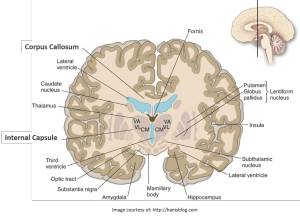 Our results suggest that white matter tracts within the developing brain have different vulnerabilities. The internal capsule, a white matter tract carrying information about the movement and sensation of the face and body, may be able to readily regenerate and repair itself. In contrast, the corpus callosum, a white matter tract connecting the two halves of the brain, was highly sensitivity to damage from the expanding ventricles and did not show signs of repair. In our study, the visual system was the most resilient to the damage caused by hydrocephalus as seen by DTI. Our DTI findings, in a clinically-relevant model of obstructive hydrocephalus, demonstrate the profound but regional effects of ventriculomegaly on white matter tracts. Future studies need to determine exactly how the white matter tracts were damaged and in some cases repaired on a cellular level.
Our results suggest that white matter tracts within the developing brain have different vulnerabilities. The internal capsule, a white matter tract carrying information about the movement and sensation of the face and body, may be able to readily regenerate and repair itself. In contrast, the corpus callosum, a white matter tract connecting the two halves of the brain, was highly sensitivity to damage from the expanding ventricles and did not show signs of repair. In our study, the visual system was the most resilient to the damage caused by hydrocephalus as seen by DTI. Our DTI findings, in a clinically-relevant model of obstructive hydrocephalus, demonstrate the profound but regional effects of ventriculomegaly on white matter tracts. Future studies need to determine exactly how the white matter tracts were damaged and in some cases repaired on a cellular level.
I have recently started my own laboratory at the Medical University of South Carolina, where we are starting to study a very important question, which up to now, has remained a mystery. How do the progenitor cells of the developing brain (those that will go on to make the billions of brain cells needed for normal development) react to the pressures exerted on the brain in neonatal and infantile hydrocephalus? Our goal will be to determine what molecular signals are released when cells are being damaged by high pressure. If we can identify and isolate the active agents/molecules that signal brain injury in the most primitive of brain cells, we can potentially use these as a therapeutic target as well as a screening tool to determine when a child’s brain is in danger or being damaged. The implications for the findings of this research are far-reaching and will hopefully guide us ever closer to the goal of our lab, which is to FIND A CURE for hydrocephalus!
