Shunt Systems
What is a Shunt?
The most common treatment for hydrocephalus is the surgical placement of a medical device called a shunt that drains the excess fluid from the brain to another part of the body.
The shunt tubing, or catheter, runs underneath the skin and ends in another part of the body where the fluid can be absorbed naturally. By doing this, the pressure that is being put on the brain is relieved.
Research has found that a shunt system is a successful treatment option, although you will need it for the rest of your life. You will also need to be monitored regularly throughout your lifetime, so finding a neurosurgeon you trust is vital.
How is a Shunt Placed?
To lower the pressure back to a normal range, a shunt is surgically placed within the brain’s ventricles. Typically a shunt is placed in the lateral ventricles (located in the center of the brain where the CSF fluid is produced), but can also be placed in the third or fourth ventricles.
A small hole is drilled into the skull and a tube, called the proximal catheter, is gently guided through the brain into the ventricle. The tube is connected to a valve typically placed behind the ear. The tubing and valve are under the skin and hair, so typically unseen. This valve mechanism regulates pressure by controlling how much fluid is allowed out of the brain to flow through the shunt tubing.
A second tube, called a distal catheter, is connected to the valve and continues to run under the skin, directing the fluid to where the fluid will be absorbed, such as the abdominal cavity, heart, or another determined area of the body.
What Are the Most Common Shunt Systems?
Below are the four types of shunt systems:
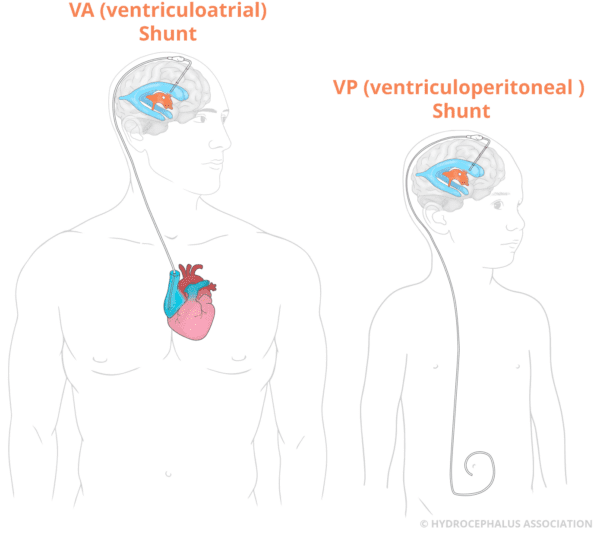 VP (ventriculoperitoneal ) shunts divert fluid from the brain’s ventricles into the peritoneal cavity, the space in the abdomen where the digestive organs are located.
VP (ventriculoperitoneal ) shunts divert fluid from the brain’s ventricles into the peritoneal cavity, the space in the abdomen where the digestive organs are located.
VA (ventriculoatrial) shunts divert fluid from the brain into the heart by placing the distal catheter into a vein in the neck, then gently advancing fluid into the right atrium.
VPL (ventriculopleural) shunts divert excess brain fluid into the pleural (chest) cavity, which is a space between the chest wall and the lungs. It is lined by a membrane along the chest wall (parietal pleura membrane) and the lung surface (visceral pleura membrane) with pleural fluid in between.
LP (lumboperitoneal) shunts are a unique type of shunt as they divert fluid from an area in the spine (called the intrathecal space) rather than the brain. This fluid is then led to the cavity within the body that houses the digestive organs.
What are the Different Types of Shunt Valves?
Every shunt has a valve. There are currently two types of valves: fixed and adjustable (programmable).
The shunt is connected to a valve that helps to regulate how much fluid leaves the brain. Most pressure control valves function on the basis of change in differential pressure (DP)-the difference between the pressure at the proximal catheter tip (in the brain) and the pressure at the distal catheter tip (in the area where the fluid is absorbed).
Neurosurgeons select a DP valve based upon:
- your age,
- the size of your ventricles,
- the amount of pressure that needs to be relieved,
- and other important clinical factors.
Fixed pressure valves regulate the pressure within the brain (called intracranial pressure or ICP) by using a one-way valve that drains to a defined intracranial pressure amount. These are implanted with a predetermined setting, meaning that they cannot be changed without additional surgery.
Adjustable (programmable) valves regulate the ICP based on a pressure setting, like the fixed pressure valve, but the setting can be adjusted by your doctor using an external adjustment tool. This allows your health care professional to non-invasively change the valve pressure setting during an office visit.
Adjustable valves are manipulated by a strong magnetic field found in the adjustment tool and, therefore, some of these valves may be sensitive to environmental magnets.
NOTE: Care must be taken to keep toys with magnets and other sources of magnetic fields away from the implanted device. It’s important to ask your doctor what precautionary measures should be taken.
The following table shows examples of Shunt Valves
To see the full product listing for each company, click on their logo.
 |
 M.blue™ Adjustable Valve M.blue™ Adjustable Valve |
 |
 Codman® Certas™ Plus Codman® Certas™ Plus |
 |
 OSV II® Flow Regulating Valve OSV II® Flow Regulating Valve |
 |
 Strata Adjustable Valve Strata Adjustable Valve |
 |

Novus™ Valve |
 Polaris Adjustable Valves Polaris Adjustable Valvesnumerous configurations available |
|
What Else Should I Know About Shunts?
What is a Reservoir?
Many valves also have a bulb-like reservoir and serve important functions.
It can be used to remove samples of CSF for testing, your doctor can inject fluid into the system to test the flow and function of the shunt, and it can be used to measure pressure. The reservoir can be felt through the skin and pumped manually, however, you should not pump your reservoir unless explicitly instructed to do so by your doctor.
These reservoirs can give insight into the shunt’s efficacy as many issues can be diagnosed by touch. In general, if one pushes on the reservoir and it does not spring back, then there might be an obstruction. If the reservoir feels rather stiff and more force is needed, this means that the valve and/or catheter may be clogged.
Can Shunts Overdrain?
In some cases, standing or sitting may cause a siphoning effect, which essentially “pulls” fluid.
Ideal hydrocephalus shunt pressure-flow functions will match your specific needs both when lying down and when standing/sitting. However, some experience a siphoning effect which results in over-drainage of CSF from the brain.
In order to prevent this from happening, the clinician may decide to add a control device to your shunt system in order to prevent over-drainage. These are called anti-siphon or anti-gravitational devices. Some of these devices are connected to the valve while some can sit further down on the distal tubing.
How do you know if fluid is flowing through your shunt system?
Your shunt drains fluid from your brain to another part of your body, but how do you know if fluid is flowing? There are two medical devices that can check to see if fluid is passing through your shunt tubing non-invasively and without the use of needles or imaging (MRI or CT Scan).
ShuntCheck by NeuroDX uses temperature to check shunt flow. A sensor is placed on the collar bone directly over the shunt tubing and it collects skin temperature data. Then a cold pack is placed on the neck just “upstream” of the sensor, chilling the shunt fluid right through the skin. If the shunt is flowing, the now cooled fluid moves downstream to the sensor and registers as a temperature drop, indicating flow. If the shunt is not flowing, the cooled fluid remains upstream, so no drop in temperature will be recorded.
FlowSense by Rhaeos, Inc. is a noninvasive wireless, wearable skin patch that can assess and monitor fluid flow beneath the surface of the skin. A small flexible wireless sensor is placed on your skin near your collar bone. The sensor sends information to an iPad. An automated flow measurement is completed in about ten minutes and the sensor is removed and discarded. Based on the flow measurement report, doctors can see if there is flow through the shunt.
Does A Shunt Need to be Flushed?
Typically, no. Additionally, standard valves should only be pumped by a medical professional.
 However, a new device is being used by some medical professionals to flush an existing shunt system which is providing a non-surgical means to restore or increase CSF flow in a non-flowing shunt. The ReFlow™ Ventricular System by Anuncia Medical, Inc. offers a qualified neurosurgeon a non-invasive means to flush the ventricular catheter in the case of a suspected blockage within the tubing. Some neurosurgeons have also found that routine flushing of the Reflow when the shunt system is working may also help to prevent a blockage before it begins. A trained individual can pump the Reflow™ Flusher which sends a controlled and limited retrograde pulse of fluid through the shunt tubing. Flushing a Reflow either to open an obstructed catheter or to prevent obstruction needs to be overseen and instructed only by a qualified neurosurgeon. The Reflow™ Flusher is compatible with any commercially available flow-regulating CSF shunt valve and peritoneal catheter. It must be placed by a qualified neurosurgeon.
However, a new device is being used by some medical professionals to flush an existing shunt system which is providing a non-surgical means to restore or increase CSF flow in a non-flowing shunt. The ReFlow™ Ventricular System by Anuncia Medical, Inc. offers a qualified neurosurgeon a non-invasive means to flush the ventricular catheter in the case of a suspected blockage within the tubing. Some neurosurgeons have also found that routine flushing of the Reflow when the shunt system is working may also help to prevent a blockage before it begins. A trained individual can pump the Reflow™ Flusher which sends a controlled and limited retrograde pulse of fluid through the shunt tubing. Flushing a Reflow either to open an obstructed catheter or to prevent obstruction needs to be overseen and instructed only by a qualified neurosurgeon. The Reflow™ Flusher is compatible with any commercially available flow-regulating CSF shunt valve and peritoneal catheter. It must be placed by a qualified neurosurgeon.
Are Shunts Visible?
Shunt systems (the valve and tubing) are placed under the skin. The valve typically sits on the head and is covered by your hair. If you have short hair, of course, your scar and valve may be visible.
It’s true, the placement of a shunt system leaves a bump under the skin. Some surgeons, however, use a cranial implant casing to securely position the valve, allowing the valve to be even with the cranium. This eliminates the bump. It may also help avoid stretching and thinning of the skin over time. The InvisiShunt® by Longeviti is a single-use sterile implant with various shapes and sizes that can encase most current shunt valves on the market. This casing is currently being used in adults.
What Are Some Complications with Shunts?
Shunts save lives and are successful in reducing the pressure in the brain for most people. A shunt allows individuals to live full lives but is not without complications, some life-threatening.
An estimated 50% of shunts in the pediatric population fail within two years of placement and additional neurosurgical operations are often required. The most common shunt complications are malfunction and infection.
For more detailed information, visit Complications of Shunt System and Preparing for Shunt Surgery.
This article was produced by the Hydrocephalus Association, copyright 2023. We would like to thank the following individuals for their valuable contributions and expert input: J. P. McAllister, PhD, Daniel J. McCusker M.S., Marion L. (Jack) Walker, MD, and Marvin Sussman, PhD.
We also would like to credit our industry partners for their continued support: Medtronic, Codman Specialty Surgical, an Integra LifeSciences Company, Aesculap, Inc., Sophysa, Natus
