New Theories in Posthemorrhagic Hydrocephalus
By: Aparna Srinivasan and Dr. Jenna Koschnitzky, Director of Research Programs
 In our last installment, we described a theory on how the disruption of the ventricular zone (VZ), the tissue surrounding the ventricles, may lead to fetal-onset hydrocephalus. Recent research suggests that similar ventricular zone disruption also occurs after intraventricular hemorrhage (IVH), a type of brain bleed, and thus may have a role in the development of posthemorrhagic hydrocephalus (PHH).
In our last installment, we described a theory on how the disruption of the ventricular zone (VZ), the tissue surrounding the ventricles, may lead to fetal-onset hydrocephalus. Recent research suggests that similar ventricular zone disruption also occurs after intraventricular hemorrhage (IVH), a type of brain bleed, and thus may have a role in the development of posthemorrhagic hydrocephalus (PHH).
Intraventricular hemorrhage (IVH) is the most common neurological complication in preterm infants. Ten percent (10%) of the infants with IVH go on to develop PHH. The risk of developing PHH increases in infants with severe brain bleeds. The placement of a ventriculoperitoneal shunt is the primary long term treatment of PHH. However, even after placement of a shunt, infants with PHH often still develop significant intellectual and motor disabilities. In addition, school aged children can have attention and academic difficulties.
Despite its prevalence and significant effect on the brain, the development of PHH is poorly understood by scientists. The predominant view amongst clinicians and scientists is that as blood accumulates and flows through the ventricular system it blocks CSF flow and damages the tissue at the site of CSF absorption. By physically blocking the flow of CSF and stopping the reabsorption of CSF, the blood causes PHH.
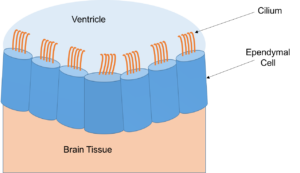
However a new theory proposes that the blood also damages the cells that line the ventricles, weakening the walls of the ventricles, and that this damage contributes to the development of PHH. The cells lining the ventricle are called ependymal cells and are part of the ventricular zone. The ependymal cells have microscopic hair-like structures known as cilia. Studies have shown that the movement of the cilia plays an important role in the movement of CSF.
In a recent paper published in the Journal of Neuropathology and Experimental Neurology, Dr. James ‘Pat’ McAllister from the Washington University School of Medicine in St. Louis, Missouri and an international team of colleagues provided data that supports this new theory. In the study, Dr. McAllister and his team of researchers studied brain samples of premature infants with IVH and premature infants who did not develop IVH (control group). They found that ventricular zone disruption, and loss of ependymal cells, occurred in the premature infants with IVH but not in the control group.
Although more studies are needed, understanding how the ependymal cells are damaged could help researchers find ways to stop or repair the damage. This could prevent or minimize the development of PHH and improve cognitive outcomes.
Dr. McAllister is a staple in the hydrocephalus research community, and his research has been supported by the Hydrocephalus Association (HA) Established Investigator Award and Innovator Award. Dr. McAllister is a member of the HA Medical Advisory Board and a long time presenter at the National Conference on Hydrocephalus. In addition, Dr. McAllister has been, and continues to be, a mentor to many graduate students, post-doctoral fellows, and medical fellows who have continued on to conduct important research on hydrocephalus.
