New Theories in Fetal-Onset Hydrocephalus
By: Noriana Jakopin and Dr. Jenna Koschnitzky, Director of Research Programs.
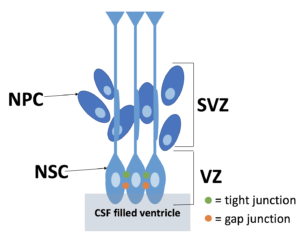 In previous blogs we explained the functions of cell junctions as well as the ventricular zone (VZ) and subventricular zone (SVZ). Current research suggests that a cascade of events involving cell junctions, the VZ, and the SVZ may be the cause of fetal-onset hydrocephalus and its accompanying neurological disorders.
In previous blogs we explained the functions of cell junctions as well as the ventricular zone (VZ) and subventricular zone (SVZ). Current research suggests that a cascade of events involving cell junctions, the VZ, and the SVZ may be the cause of fetal-onset hydrocephalus and its accompanying neurological disorders.
The neural stem cells (NSCs) make up the VZ and eventually differentiate into ependyma, the cells lining the ventricles. NSCs and ependyma are held together by different types of junctions. As a reminder, gap junctions use proteins called connexins and provide a channel for cell to cell communication when cells are close together, but not physically touching each other. Adherens junctions use proteins called cadherins and anchor cells to one another by providing a stiff connection between the cells. Tight junctions use proteins called claudins that create a barrier that does not allow fluid to pass in between the two cells.
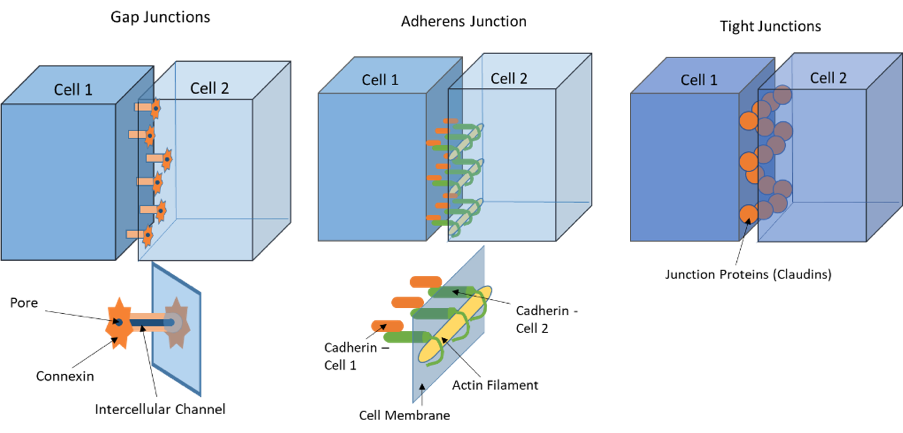
In order for these junctions to function properly, it is essential that the correct proteins are deposited in the correct location. However, research suggests that in fetal-onset hydrocephalus, the cellular transport mechanisms which deposit the essential proteins to make the gap and adherens junctions are disordered. Several different genetic mutations can affect this transport process, causing the misplacement of proteins. This results in cell junctions which are unable to function properly, thus disrupting the connections between cells. The resulting cell junction abnormalities cause a disruption to the structure of the VZ and result in the loss of VZ cell layer in certain areas.
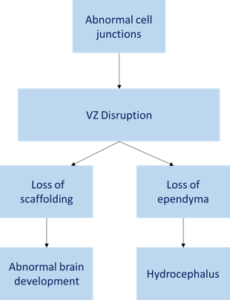 VZ disruption occurs in a very specific spatial and temporal pattern during fetal development. VZ disruption in the cerebral aqueduct results in the absence of ciliated ependyma in the aqueduct which leads to aqueductal stenosis, or narrowing of the aqueduct, and hydrocephalus.
VZ disruption occurs in a very specific spatial and temporal pattern during fetal development. VZ disruption in the cerebral aqueduct results in the absence of ciliated ependyma in the aqueduct which leads to aqueductal stenosis, or narrowing of the aqueduct, and hydrocephalus.
VZ disruption in the telencephalon, the fetal precursor to the cerebrum, causes NSCs and neural progenitor cells (NPCs) to fall into the CSF. Since the maturation of NPCs requires the use of NSCs as scaffolding, any NPCs that have not been lost to CSF are unable to properly mature and begin migration. Thus, the VZ disruption alters the pathways of neurogenesis, resulting in abnormal brain development.
There has been recent research in support of this theory on the progression of fetal-onset hydrocephalus and its accompanying neurological challenges. Recently, Dr. Esteban M. Rodriguez and Dr. Maria M. Guerra delineated the theory in a review paper based on such research. They believe that this research opens the possibility for the development of therapies using NSCs and offers an explanation as to why the treatment of hydrocephalus does not reverse many of the associated neurological disorders (e.g. epilepsy).
