New Study Seeks to Improve Outcomes After a Brain Bleed
By: Maggie A. Khuu and Jenna Koschnitzky, Ph.D., Director of Research Programs
Germinal matrix hemorrhage (GMH) is a brain bleed that occurs in approximately 3.5 per 1000 live births and remains a leading cause of mortality and lifelong morbidity in premature infants. A common occurrence that arises after GMH is the development of subsequent complications such as developmental delays, learning and psychiatric disorders, and posthemorrhagic hydrocephalus (PHH). While the focus has been primarily concentrated on preventing GMH from occurring by preventing preterm birth, very little has been studied on how to non-invasively prevent complications, such as PHH, once GMH has already occurred.
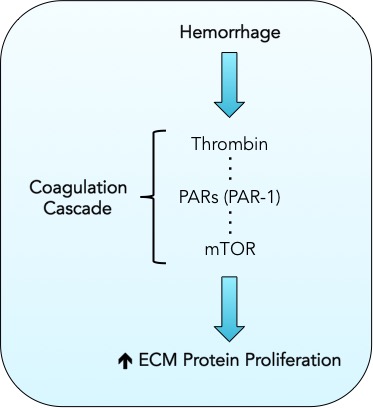
Researchers from Loma Linda University sought to prevent the development of PHH in an animal model through the use of dabigatran, an oral anticoagulant, following GMH induction. Immediately following a brain bleed, a coagulation, or blood clotting, signaling cascade occurs and is associated with the increase in production of extracellular matrix (ECM) proteins in fibroblasts (see figure). ECM proteins lead to a buildup of scar tissue in the brain and are hypothesized to disrupt cerebral spinal fluid dynamics in the brain. This disruption, in turn, may give way to the development of PHH. Dabigatran works by inhibiting thrombin, one of the enzymes that gets activated during the coagulation cascade.
In this study, researchers from the Zhang lab used dabigatran to prevent thrombin production. As an alternative, they also looked at preventing one of thrombin’s downstream receptors, PAR-1. Given dabigatran’s anticoagulant properties, this alternative was tested to see if the same therapeutic benefits could be achieved through a different route. What they found was that while thrombin inhibition improved long-term neurofunctional recovery, long-term brain morphological outcomes, and significantly reduced long-term ECM proliferation, PAR-1 inhibition alone was not sufficient enough to be effectively address these issues. These findings indicate that thrombin activity is associated with posthemorrhagic hydrocephalus development and that thrombin induced PAR-1 activation plays a minor role in conjunction with other activated pathways. This suggests that future studies investigating post-GMH hydrocephalus development should delve more into alternative pathways as they may illuminate the other options not yet explored in ECM protein proliferation.
John H Zhang, MD, PhD, the senior author in this study and member of Hydrocephalus Association Network for Discovery Science (HANDS), spoke at the Hydrocephalus Association’s Posthemorrhagic Hydrocephalus workshop this past July.
