HA Established Investigator Publishes Research Findings on Congenital Hydrocephalus

by Ashly Westrick, Research Programs Manager
James P. (Pat) McAllister, Ph.D., is the Director of Basic Hydrocephalus Research at the University of Utah. In 2011, he received a two-year Established Investigator grant from the Hydrocephalus Association. The journal article, The role of the subcommissural organ in the pathogenesis of congenital hydrocephalus, recently published in Cell Tissue Research, details his research findings.
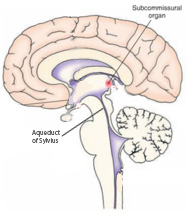
The subcommissural organ (SCO) is located at the entrance of the aqueduct of sylvius (SA) and is highly specialized in the secretion of proteins. In 1954, Overholser et. al. hypothesized that these proteins prevented the closure of the aqueduct of sylvius and that maldevelopment of the SCO could lead to stenosis (or narrowing) of the aqueduct and, consequently, hydrocephalus. Multiple studies have supported this hypothesis. However, it is unknown whether the hydrocephalus precedes or results from the maldevelopment of the SCO. The current study provides evidence that maldevelopment of the SCO precedes hydrocephalus, indicating that it is possible the maldevelopment is causing the hydrocephalus. The researchers compared HTx rats to normal rats. HTx rats are a mutant rat that presents with fetal-onset hydrocephalus with stenosis of the SA and dilation of the lateral ventricles starting at embryonic day 18 (E18). The researchers discovered abnormalities in the subcommisural organ as early as embryonic day 15 in the HTx rat, indicating that maldevelopment of the SCO occurs prior to hydrocephalus.
 Furthermore, the subcommisural organ secretes glycoproteins, most of which aggregate and form Reissner’s fibers (RF). There is evidence that the Reissner’s fibers may be involved in the regulation of the composition of the cerebrospinal fluid (CSF). In non-affected rats, the central canal of the spinal cord displays RF running the whole length. Hydrocephalic rats were devoid of RF. The lack of these fibers could have two possible consequences. First, it is hypothesized that the proteins secreted by the subcommisural organ provide a mass of negatively charged molecules that help to keep the aqueduct open. Without these proteins, there is a diminished amount of negative charges in the SA and this contributes to a narrowing of the aqueduct. Secondly, evidence suggests that the fibers are a key element for normal CSF flow through the aqueduct. The absence of RF could impact CSF hydrodynamics.
Furthermore, the subcommisural organ secretes glycoproteins, most of which aggregate and form Reissner’s fibers (RF). There is evidence that the Reissner’s fibers may be involved in the regulation of the composition of the cerebrospinal fluid (CSF). In non-affected rats, the central canal of the spinal cord displays RF running the whole length. Hydrocephalic rats were devoid of RF. The lack of these fibers could have two possible consequences. First, it is hypothesized that the proteins secreted by the subcommisural organ provide a mass of negatively charged molecules that help to keep the aqueduct open. Without these proteins, there is a diminished amount of negative charges in the SA and this contributes to a narrowing of the aqueduct. Secondly, evidence suggests that the fibers are a key element for normal CSF flow through the aqueduct. The absence of RF could impact CSF hydrodynamics.
Another finding was that transthyretin (TTR) is present in higher a concentration in HTx rats than normal rats. TTR is a protein involved in the transport of thyroid hormones in the CSF. The main source of this protein in CSF is the choroid plexus. The choroid plexus of the hydrocephalic rat probably secretes more TTR than in the normal rat. This indicates that factors that modulate TTR levels in hydrocephalus CSF could be important to consider in hydrocephalus.
The ultimate hypothesis from the research is that the absence of the large mass of negatively charged molecules from the aqueduct of sylvius results in a narrowing of the aqueduct, impairs the flow of CSF through the aqueduct, and causes severe hydrocephalus.
More research is needed to understand the genetics of this mechanism but this paper provides valuable insight into the role of SCO in congenital hydrocephalus.
Click here to read the abstract of the paper.
REFERENCE:
OVERHOLSER, M. D., WHITLEY, J. R., O’DELL, B. L. & HOGAN, A. G. (1954). Ventricular system in hydrocephalic rat brains produced by deficiency of vitamin B12 or folic acid in maternal diet. Anat. Rec. 120, 917.
