CSF Shunt Entry Site Trial
The CSF Shunt Entry Site Trial has reached 75% enrollment!

The CSF Shunt Entry Site Trial has reached the 75% mark in patient enrollment – that is 385 patients enrolled! Lead investigator, Dr. William Whitehead, expects to complete enrollment by December 2018.
A huge THANK YOU to all of the families involved with this trial!
About the trial:
In 2014, the Patient-Centered Outcomes Research Institute (PCORI) awarded a $1.8 million contract in support of a Hydrocephalus Clinical Research Network (HCRN) randomized control trial to determine the most effective entry site for placing a shunt. The Hydrocephalus Association (HA) partnered with HCRN to involve patients and caregivers in the initial study selection and development.
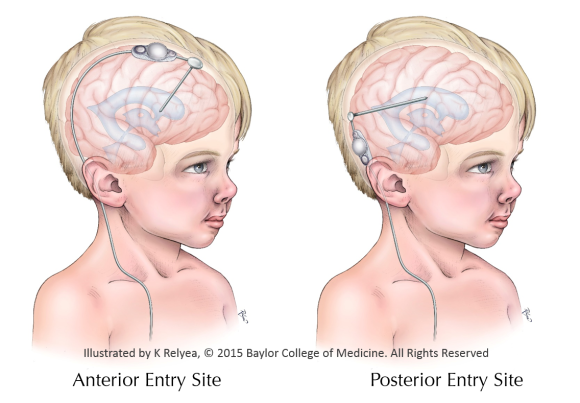
The most common treatment of hydrocephalus is the placement of a shunt. There are two possible entry sites which surgeons use to place the shunt, but it is unclear which, if either, entry site is better. The CSF Entry Site Trial is using randomized trial methods to compare the two possible entry sites.
About PCORI:
The funding organization, PCORI, is a nonprofit, nongovernmental organization established by Congress under the Patient Protection and Affordable Care Act of 2010. PCORI aims to fund research that is most relevant to patients and its funded contracts require involvement by the patient community throughout the entire lifecycle of a project. To learn more about how PCORI is putting patients at the center of research CLICK HERE.
