Rhaeos CEO Shares Details About Wearable FlowSense Device
 Earlier this year, Rhaeos won MedTech Innovator's 2020 Global Competition. The award will help further the development of their wearable, noninvasive FlowSense device. In a recent conversation with Rhaeos CEO Anna Lisa Somera, Somera discussed the significance of the award and how FlowSense can improve the way hydrocephalus is managed.
Earlier this year, Rhaeos won MedTech Innovator's 2020 Global Competition. The award will help further the development of their wearable, noninvasive FlowSense device. In a recent conversation with Rhaeos CEO Anna Lisa Somera, Somera discussed the significance of the award and how FlowSense can improve the way hydrocephalus is managed.
Congratulations on winning the MedTech Innovator's 2020 Global Competition award! What’s the significance of this award?
MedTech Innovator (MTI) is the largest global medical device-only accelerator and is sponsored by big industry players such as Johnson and Johnson, Olympus, Nipro, Baxter, and more. With the win, we took home $350,000 in cash plus Johnson and Johnson Labs (JLabs) space. This year, MTI had over 1,000 applicants and taking the top spot has been an enormous honor. It is the culmination of a great deal of hard work from the Rhaeos team in collaboration with a fantastic slate of advisors. Our biggest goal when participating in MTI was to really listen to the advisors and incorporate as much of their knowledge and wisdom as possible into our strategic plans. But to win the Grand Prize was beyond our wildest imagination. Additionally, to be the first female to take the top spot this makes the win even more meaningful.
What is FlowSense and how does it work?
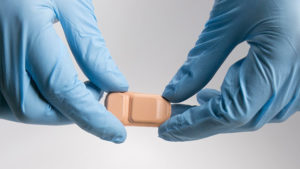
FlowSense is a wireless, non-invasive thermal flow sensor that affixes to a patient’s neck, overlying the shunt used to relieve cerebrospinal fluid (CSF) pressure, where it detects the presence and magnitude of CSF flow. The device uses a small heater to gently increase the temperature of the skin directly above the shunt tubing and then precisely measures that temperature at upstream and downstream locations. When there’s no flow, we’ve seen that the temperature of the skin increases isotropically - equally in all directions around the heater. But when there is flow, the heat moves anisotropically – unequally around the heater – with a small but measurable relative temperature increase downstream from the heater. We use high-precision temperature sensors and Bluetooth technology to send this temperature data to an iPad, which analyzes the flow and gives feedback to the physician. This is all done with a small flexible device with a form factor that makes it look a lot like a bumpy adhesive bandage. With FlowSense, we can monitor shunt function in outpatient clinics, inpatient settings, emergency departments, and patients’ home. Our company aims to significantly reduce unnecessary imaging, hospital length of stay, and readmission costs.
Where did the idea for this device come about?
The idea behind the FlowSense device came about when Rhaeos Co-founder, Dr. John Rogers, and Northwestern Memorial Hospital neurosurgeon, Matthew Potts, connected to discuss the use of sensors as a better way to monitor treatment flow in hydrocephalus patients. Dr. Rogers, a world-renowned wearables inventor, and Louis Simpson and Kimberly Querrey, Professor of Materials Science and Engineering, Biomedical Engineering and Neurological Surgery at Northwestern, was intrigued by this potential application and looped me in. I worked in medical devices for 16 years, so when I heard about this opportunity, I hit the brakes on everything I was doing, knowing that this technology would become something exceptional. Then upon taking the helm, I recruited teammates from previous start-ups to which we participated. I wanted start-up veterans with battle scars. And it has paid off, as we continually hit our milestones primarily due to the team and its passion for our product.
When do you anticipate that the product will be available clinically? Are you currently pursuing clinical trials?
Our first version of FlowSense, which we expect to bring to market in late 2021, is a single-use disposable device intended for use in the hospital. It takes only about five minutes to complete the flow assessment. In our next-generation device, which we’ve recently begun to develop with a strategic partner, patients will be able to use the device at home – much like many of us use wearable fitness trackers – to not only assess flow at a given time, but also to predict a shunt failure before it sends them to the emergency room.
To date, we’ve been thrilled to work with world-renowned academic partners who have performed the first clinical evaluations of this technology at Northwestern Memorial Hospital and Lurie Children’s Hospital in Chicago, with the work published in Science Translational Medicine and Nature Partner Journal’s Digital Medicine.
We’ve also had the privilege of working closely with the FDA to define our regulatory path and clinical data needs after having received a breakthrough device designation for FlowSense. We anticipate kicking off additional clinical studies early next year to gather data for regulatory approval and to show the economic value of FlowSense.
We anticipate that a great deal of the product development and clinical work will be funded through pending NSF and NIH grants in combination with investors in our current seed round. We plan to follow that with a Series A round to scale the device.
What is next up for Rhaeos?
As a start-up CEO, you are always fundraising. I am raising our seed round of $1.5M to get through 2021 and FDA approval. (Interested investors should reach out to Anna Lisa at info@rhaeos.com).
