Clinical Networks
The HCRN and AHCRN are focused on improving the lives of those living with hydrocephalus.
Our Clinical Networks are Making a Difference!
The HCRN and AHCRN are focused on improving the lives of those living with hydrocephalus.
Their research programs range from understanding the natural history of the condition to optimizing surgical procedures and protocols and finding ways to identify people who are at risk for developing hydrocephalus.
Treatment Options
The clinical networks are evaluating different treatment options for both our pediatric and adult populations.
Infection Prevention & Treatment
The clinical networks are working to decrease the impact of shunt infections by developing protocols that reduce the risk of shunt infections and optimizing treatment strategies when shunt infections do develop.
Shunt Longevity
The clinical networks are finding ways to increase the life of a shunt.
Cognitive Outcomes
The clinical networks are working to understand how different treatments affect long-term outcomes.
Natural History
The clinical networks are discovering how hydrocephalus affects people throughout their lives. By understanding the natural history of the condition, the networks will be able to measure changes in outcomes when new therapies are developed.

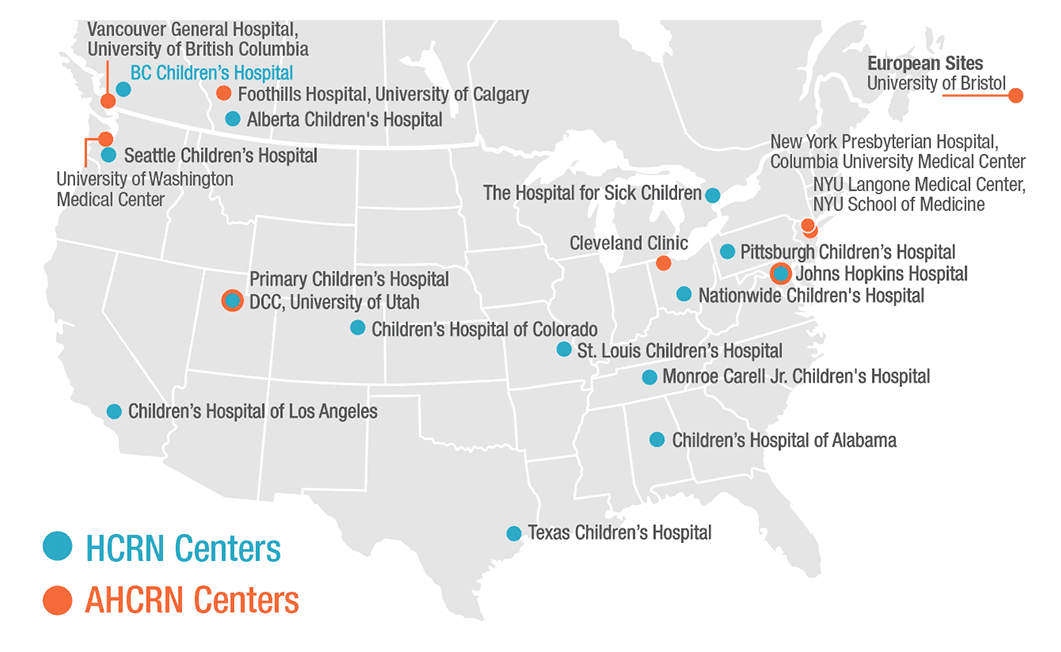
Hydrocephalus Clinical Research Network (HCRN)
A network of fourteen children’s hospitals that conduct clinical research on hydrocephalus.
Learn more at HCRN.org

Adult Hydrocephalus Clinical Research Network (AHCRN)
A network of eight hospitals that conduct clinical research focused on adults with hydrocephalus.
Learn more at www.AHCRN.org.
